Projects
TRANSITION: Delivering Innovative Materials for Medical Devices
Realising an efficacious therapeutic solution for treating osteoarthritis (OA) remains elusive and is considered one of the greatest challenges in the field of orthopaedic medicine. The overall goal of the TRANSITION spoke programme is to develop a new class of 3D printed biological implant that will regenerate rather than replace diseased joints. This hybrid device will consist of a titanium core providing mechanical integrity, overlaid by a layer of functional tissue engineered bone and articular cartilage. Realising this ambitious goal will first require addressing existing challenges in the 3D printing of metals (Aim 1), biodegradable polymers (Aim 2), bioinks and cells (Aim 3), and subsequently integrating the printing processes of these diverse material sets to develop hybrid metal-biological devices capable of restoring joint function (Aim 4). The impact of TRANSITION will be multi-faceted. Firstly, it will transform the production of metallic orthopaedic devices used in hip and knee arthoplasty, as much of the developed knowledge from this project are directly applicable to such implants. TRANSITION will also provide platform technologies to facilitate the development of new devices targeting specific clinical problems, and will ultimately enable the development of a new class of regenerative implant for treating OA.
Collaborators: Prof. Mick Morris (TCD); Prof David Hoey (TCD); Prof Conor Buckley (TCD); Prof. Fergal O’Brien (Royal College of Surgeons); Prof. Eoin O'Cearbhaill (UCD); Prof. Pieter Brama (UCD); Prof. Miles Turner (DCU); Prof Garret O'Donnell (TCD); Prof Daniel Trimble (TCD); Prof Rocco Lupoi (TCD).
Funded by: Science Foundation Ireland by a Spokes Award (17/SP/4721) and Johnson & Johnson. (Include logos)
ANCHOR: Articular cartilage regeneration through the recruitment of bone marrow derived mesenchymal stem cells into extracellular matrix derived scaffolds anchored by 3D printed polymeric supports
Osteoarthritis (OA), the most common form of arthritis, is a serious disease of the joints affecting nearly 10% of the population worldwide. The onset of OA has been associated with defects to articular cartilage that lines the bones of synovial joints. Current strategies to treat articular cartilage defects are ineffective and/or prohibitively expensive. The aim of ANCHOR is to develop and commercialise a new medicinal product for articular cartilage regeneration that recruits endogenous bone marrow derived stem cells into an extracellular matrix derived scaffold anchored to the subchondral bone by 3D printed polymeric supports. By recruiting endogenous cells into a supporting scaffold, ANCHOR will obviate the need for pre-seeding scaffolds with cells prior to implantation into cartilage defects, thereby dramatically reducing the cost and complexity of the repair procedure. It will also overcome the need for suturing of a scaffold into a cartilage defect, which is a very time consuming and technically challenging surgical procedure. Finally, the inherent chondro-inductivity of the cartilage ECM derived scaffolds developed by the applicant will maximise the potential for hyaline cartilage regeneration. The project will leverage the applicants extensive experience in ECM derived biomaterials and 3D printing to develop a new product with significant commercial potential. The impact of ANCHOR will be multi-faceted: it will transform how damaged joints are treated by orthopaedic surgeons, it will create economic value through the commercialization of IP, and most importantly it will improve patient experience and their long-term health and well-being.
Collaborators: Fergal O’Brien (Royal College of Surgeons); Prof. Pieter Brama (UCD).
Funded by: European Research Council Proof of Concept grant (779909).
CarBon: Controlling Cartilage to Bone Transitions for Improved Treatment of Bone Defects and Osteoarthritis
Many people suffer from diseases of the locomotor system, such as bone defects or osteoarthritis, for which current treatments are insufficient. Understanding and controlling the dual character of cartilage is pivotal: insufficient transition impairs bone healing, and undesired transition to bone leads to osteoarthritis. In CarBon, state of the art in vitro, in silico and in vivo models will be uniquely combined to elucidate how this transition is orchestrated and how it can be modulated. In a multifactorial approach, a network of 14 young scientists will aim to identify the biological and physical factors that determine the fate of cartilage. Knowledge from the fields of tissue engineering, cartilage and bone developmental biology and pathobiology will be combined with skills from the disciplines of cell biology, computational modelling, biotechnology (bioreactors, biomaterials) and drug discovery.
Collaborators: ERASMUS UNIVERSITAIR MEDISCH CENTRUM ROTTERDAM, KATHOLIEKE UNIVERSITEIT LEUVEN, IRCCS AZIENDA OSPEDALIERA UNIVERSITARIA SAN MARTINO, KLINIKUM DER UNIVERSITAET ZU KOELN, THE ROYAL VETERINARY COLLEGE, SurgaColl Technologies Limited, LIFETEC GROUP BV.
Funded by: Horizon 2020 / Marie Sklodowska-Curie Innovative Training Network (EU project 721432).
JointPrinting: 3D Printing of Cell Laden Biomimetic Materials and Biomolecules for Joint Regeneration.
Osteoarthritis (OA) is a serious disease of the joints affecting nearly 10% of the population worldwide. Realising an efficacious therapeutic solution for treating OA remains one of the greatest challenges in the field of orthopaedic medicine. This proposal envisions a future where 3D bioprinting systems located in hospitals will provide ‘off-the-shelf’, patient-specific biological implants to treat diseases such as OA. To realise this vision, this project will use 3D bioprinting to generate anatomically accurate, biomimetic constructs that can be used to regenerate both the cartilage and bone in a diseased joint. If successful, such an implant would form the basis of a truly transformative therapy for treating degenerative joint disease.
Collaborators: Fergal O’Brien (Royal College of Surgeons); Prof Eben Alsberg (Case Western); Prof Gordon Wallace (University of Wollongong); Prof. Pieter Brama (UCD).
Funded by: European Research Council Consolidator grant (ERC-2014-CoG-647004).
Decellularised extracellular matrix derived scaffold incorporating freshly isolated joint tissue derived cells as a bioactive implant for cartilage repair
We have developed a new ‘single-stage’ or ‘off-the-shelf’ approach to articular cartilage regeneration that consists of a chondro-inductive extracellular matrix (ECM) derived scaffold seeded with freshly isolated joint tissue derived stromal cells that are processed ‘in-theatre’. The primary objective of this project is to evaluate the efficacy of this ‘single-stage’ or ‘in-theatre’ therapy for articular cartilage regeneration in a large animal model study.
Collaborators: Prof. Fergal O’Brien (Royal College of Surgeons); Prof. Conor Buckley (TCD); Prof Kevin Mulhall (Mater Hospital & TCD); Prof Cathal Moran (Sports Surgery Clinic & TCD).
Funded by: Enterprise Ireland Commercialization Fund (CF/2014/4325).
A tissue engineered biological joint replacement prosthesis for the treatment of degenerative joint disease.
Description: Realising an efficacious therapeutic solution for treating osteoarthritis remains elusive and is considered one of the greatest challenges in the field of orthopaedic medicine. Tissue engineering aims to replace or regenerate damaged or diseased tissues through the combined use of cells, scaffolds and physio-chemical cues. The aim of this proposal is to tissue engineer a biological joint replacement prosthesis using mesenchymal stem cells (MCSs) as the basis of a novel therapy for treating degenerative joint disease. This will be achieved in part by recapitulating key aspects of normal bone and joint development, specifically the process of endochondral ossification where a hypertrophic cartilaginous intermediary is replaced by bone. The first phase of the project will attempt to tissue engineer a hypertrophic cartilaginous construct using MSCs that mimics the anatomy of the knee joint and then demonstrate that such a construct will be replaced by bone in vivo. The second phase of the project will use MSCs to engineer a stable layer of articular cartilage on the surface of the hypertrophic construct. The final phase of the project will evaluate the potential of the proposed biological prosthesis to regenerate both the articular cartilage and underlying bone of the medial femorotibial joint.
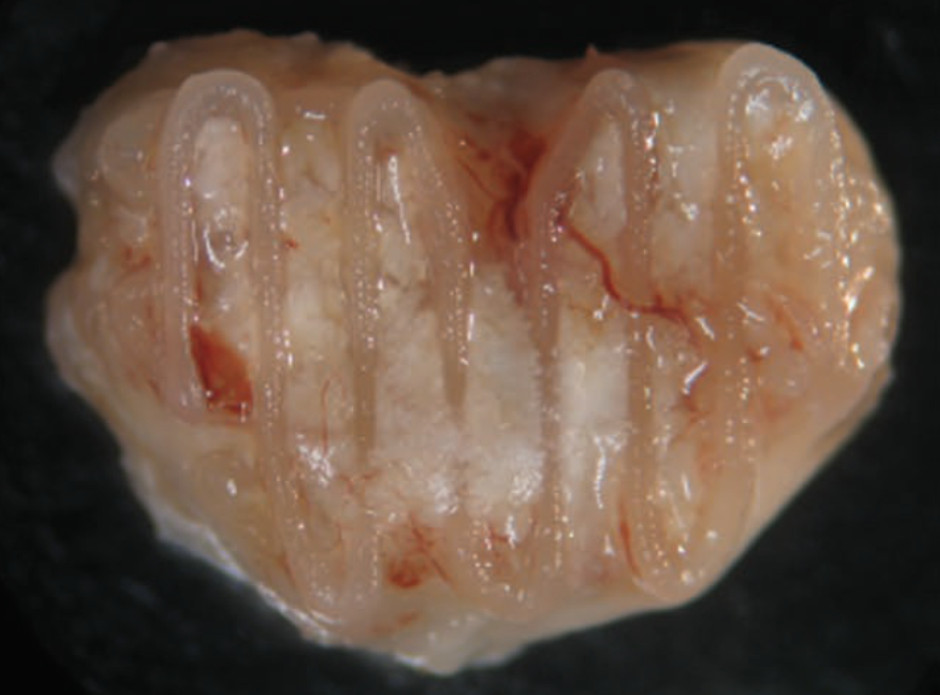
Collaborators: Prof. Fergal O’Brien (Royal College of Surgeons); Prof. Conor Buckley (TCD); Prof. Pieter Brama (UCD); Prof. Paula Murphy (TCD); Dr. Mary Murphy (NUIG); Mr Kevin Mulhall, (Consultant Orthopaedic Surgeon, Mater University Hospital).
Funded by: Science Foundation Ireland Principal Investigator Award (12/IA/1554)

